There are three types of acids: hydrochloric acid, sulfuric acid, and nitric acid. All of them dissolve in water and are electrolytes. However, nitric and sulfuric acids are less likely to completely dissolve in water. That is because they have weaker intermolecular forces than the other two. Therefore, they are more resistant to the forces of water.
A solution of these substances is acidic. An acidic substance will dissolve in water if it reacts with an equimolar amount of a base. In other words, an acid will be more soluble in water than a base. The pH of water is 7 when the two substances are in equilibrium, and when one is more electrolyte than the other, the mixture will become more electrolyte.
The second type of acid will dissolve in water if it is in an acidic state. The base will not react with the acid, which causes the solution to be a colloid. These types of solutions contain small particles that can be easily dissolved in water. A weak acid will partially dissolve in water and will be ionized when it dissolves in the aqueous medium. These two types of acids will not completely dissolve in water.
The second type of acid will not completely dissolve in water. It will remain a solid. It will remain in solution until it is neutralized with a strong acid. When this happens, it will be a polar solution. It will not dissolve in water at all. The second type will completely dissolve in water, and it will not be a polar liquid. As long as it is a dilute substance, it will not dissolve in water.
The PH of a solution is determined by the number of hydroxide ions it contains. The lower the pH, the more basic the solution is. Using the above formula, a solution of sodium chloride will dissolve in water. But a weak acid will not completely dissolve in water. The acid will be insoluble and will be a colloid. A weak acid is one that is not completely ionised.
The solubility of an acid depends on the substance. The acid can be an acid or a base, and it can be either. The stronger acid is an acid, while the weaker one is a base. If a base is an acid, it will dissolve completely in water. The other acid will completely dissolve in water if it is a weak acid. It will remain a liquid if it is a base.
The chemical equation of an acid and a base is simple. A neutral solution is one that has a pH of 7. An acid will be an acid, while a base will be a base. The latter will have a higher pH than a weak acid, and it will be the strongest acid. It will completely dissolve in water only if it has a high concentration. This is why an acid will not fully dissolve in water.
Strong acids are hydrochloric acid, while weak acids are acetic acid. The stronger acid is hydrogen chloride. The weaker one is acetic acid. Both of them do not fully ionize in water. When in water, acetic acid dissociates into hydrogen ions and acetate ions. The smallest acid will not completely dissolve in water. The strongest acid is the sulfuric and nitric acid.
A weak acid will completely dissolve in water. A strong acid will completely dissociate in water. Hence, it would be most likely to completely dissolve in water. So, which of these acids would be least likely to fully dissolve in water? The acid would completely dissociate in the water, but the weakest one would not. But it is the strongest acid. It will be the weakest. If you want to know more about acids, read the following article.









































































































































































































































































































































































































































































































































































































































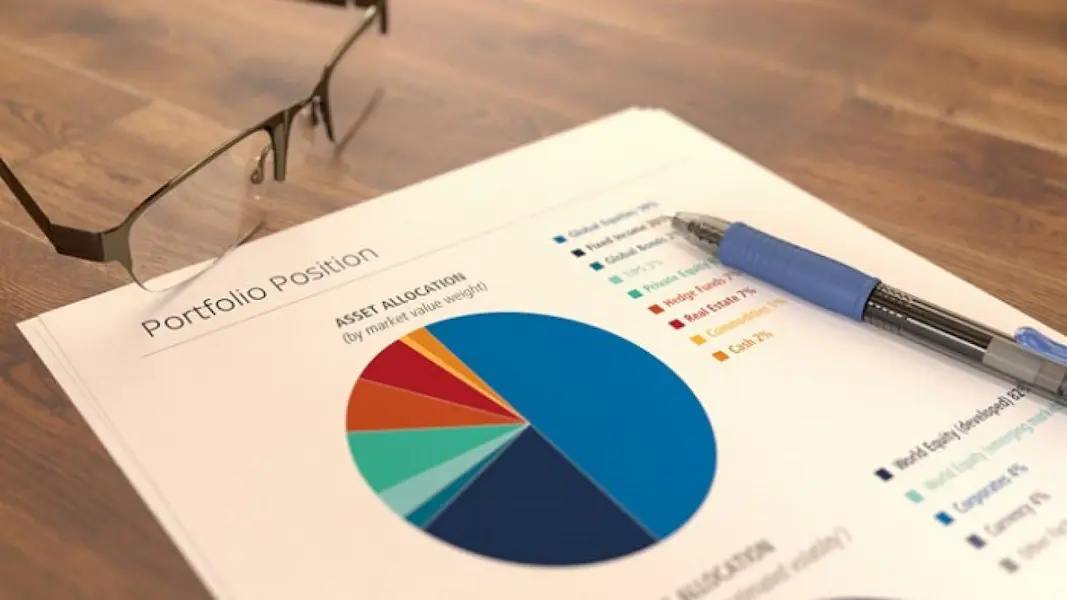








































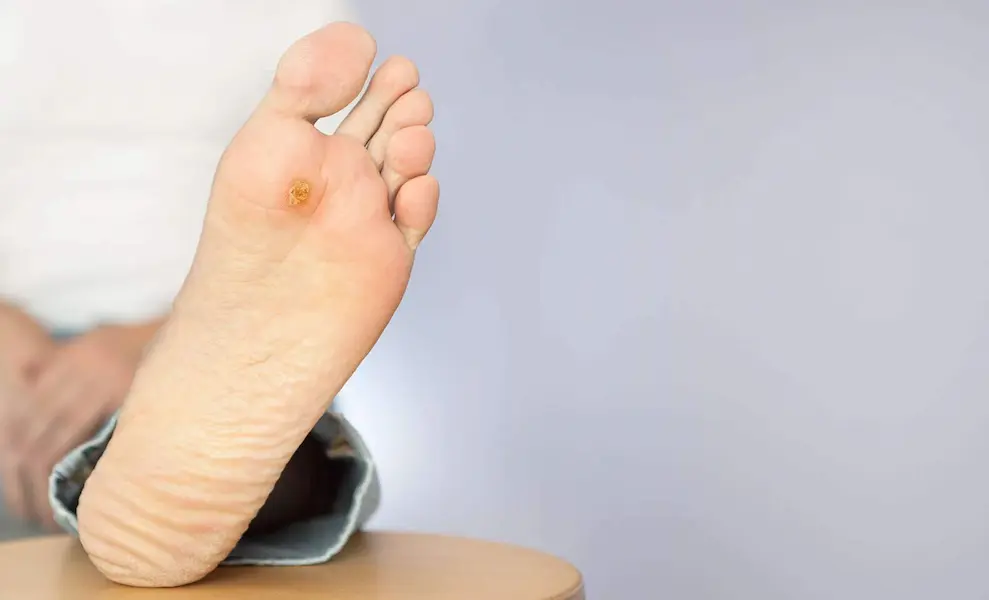

































































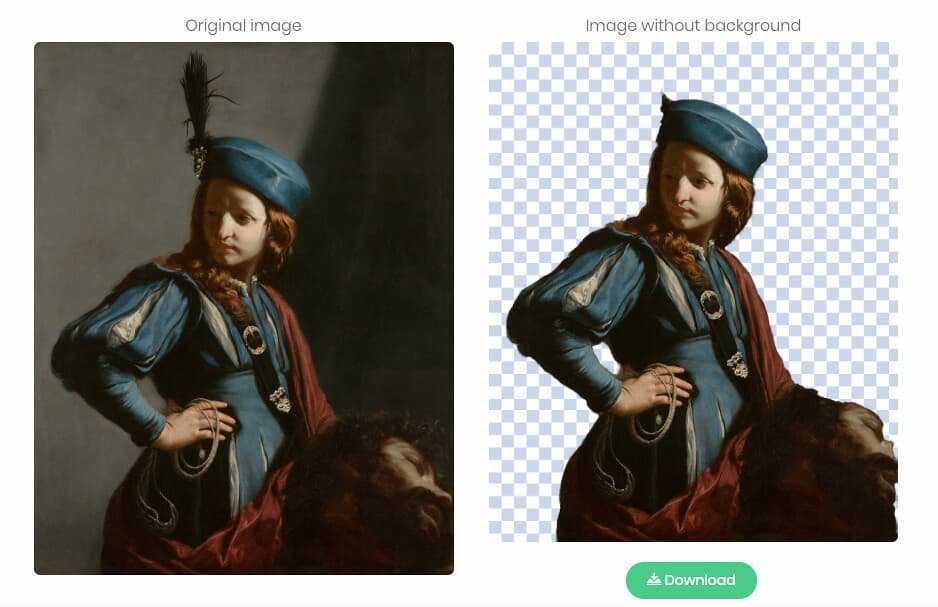






























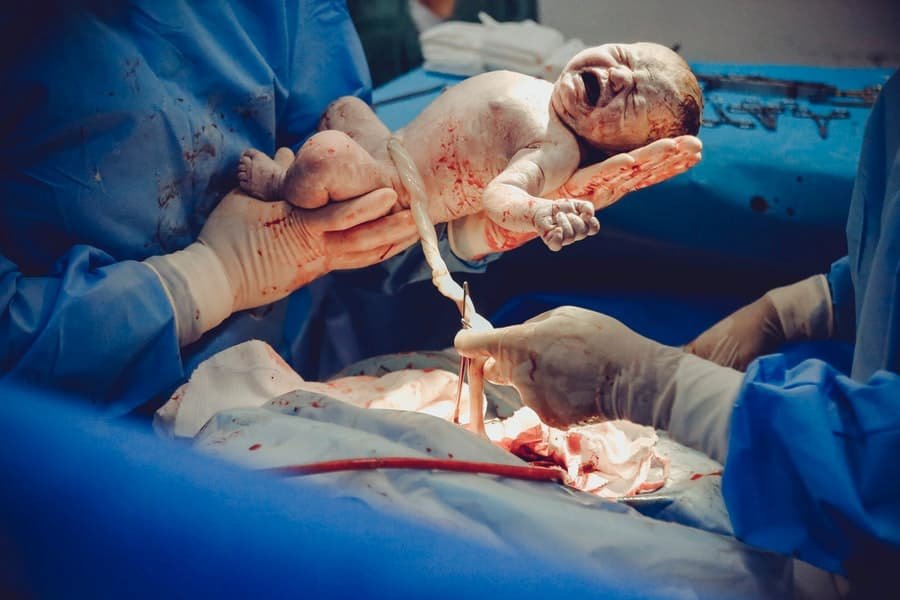






























































































































































































































































































0