Which of these statements is true about water? The ionic bond between hydrogen and oxygen holds water together. This bond is formed when hydrogen ions are too high in a solution. Hydrogen binders are formed when excess hydrogen ions form in a solution. A solution containing both acid and base will be neutralized by the other. A solution containing an acid or base should have a pH of 0 or less.
The structure of water molecules is bent. Two hydrogen atoms bond to one oxygen atom. Each hydrogen atom carries two unshared electrons. This arrangement causes water to have a polar charge. Hydrogen is more electronegative than oxygen, but oxygen has more electrons. This makes water more appealing to other substances. Water is also polar, with the positive side of the atom associated with the minus side.
The most abundant molecule in the atmosphere is water. Water makes up 60-70 percent of the human body. It is the only substance that is as abundant in the atmosphere of our planet. The existence of water on other planets and moons in the solar system means that life has been present there. Water is actually the second most abundant element of the universe. It is a necessary element for life. Its versatility as solvent is vital for living organisms.
The boiling point for water is 100 degrees Celsius. In liquid form, the hydrogen bonds form between two water molecules, causing them to become closer together. Water molecules in solid form have a lower density that their liquid counterparts. It is therefore less dense than water and can float on water’s surface. The volume of matter in liquids determines their density. Which of these statements is true about water?
Water’s heat capacity allows it to absorb great amounts of heat. This property allows water, unlike land, to cool down more slowly. Because water has the highest heat potential of all liquids, oceans cool off slower than land. Water helps heat-loving organisms evenly disperse heat. It acts like a car cooling unit, keeping their internal temperatures controlled. In the end, water makes life possible for humans.
Pure water contains 1 x 10-7 moles of H+ ions per Liter. The pH of water is the negative logarithm of hydrogen ions. The pH of water is 7.0. This is the base-base reaction. One mole of water is equal to 6.02 x 1023 molecules. You can observe water “climbing up” a tube if you are watching a similar process.







































































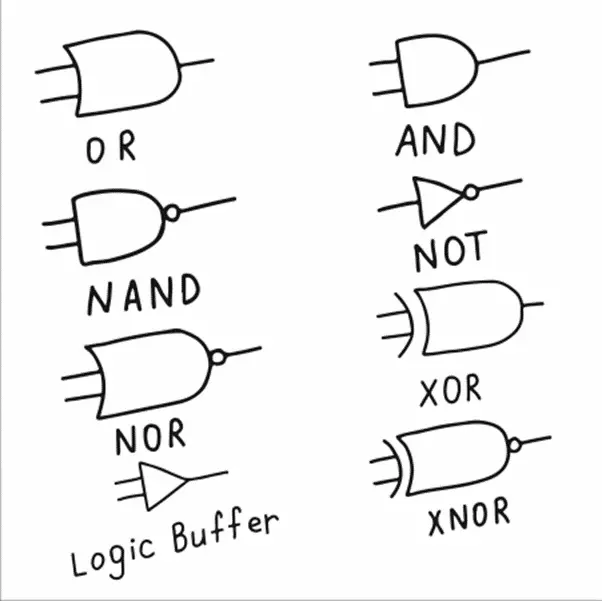







































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































0