In chemistry, there are two main descriptions for gases: ideal and real. The ideal gas constant is defined as the quantity of a gas that has no fixed volume or shape. In comparison to other states of matter, gases are much lighter than other solids and have a high proportion of empty space between particles. The kinetic energy of the molecules diffuses throughout the gas container and creates an equilibrium between the particles.
Unlike liquids and solids, gases are characterized by their varying volumes. This is because they contain a high number of molecules. The volume of a gas is directly proportional to the number of molecules. Therefore, the volume and pressure of a gas will always change correspondingly. However, a gas has a higher temperature than its ideal counterpart. For this reason, it is more common to think of a real-world gas than an ideal-gas, and vice versa.
Nevertheless, a real gas is more dense than an ideal one, and the two descriptions are not equal. If we take helium, the volume is greater than that of its ideal counterpart. To solve this problem, we can use Avogadro’s Law. This law requires that a certain mass of a real gas is converted into a number of moles, which is the equivalent of a kilogram.
The volume and pressure of a gas are directly proportional to the number of molecules in it. This is why it’s impossible to convert a real gas to an ideal one. The pressure of a real gas is lower than the pressure of an ideal one, and this difference in pressure can lead to a large difference in its volume. Avogadro’s Law can solve this problem by converting grams of Helium gas into moles.
The volume of a real gas is larger than that of an ideal one. This problem can be solved by using Avogadro’s Law. It states that the product of volume and pressure is constant. This law is used to determine the volume of a gas. Hence, you must convert the mass of Helium into a mole. This equation is the same as that of the ideal gas.
Volume and pressure are directly proportional to the number of molecules present in a gas. The volume of a real gas is larger than the volume of an ideal one. Avogadro’s Law states that the product of volume and pressure is the same. You can find the same value of volumes and pressure by combining the two. If you are uncertain about which description is best for a specific material, consider the inverse formula for determining the mass.
Visit the rest of the site for more useful articles!































































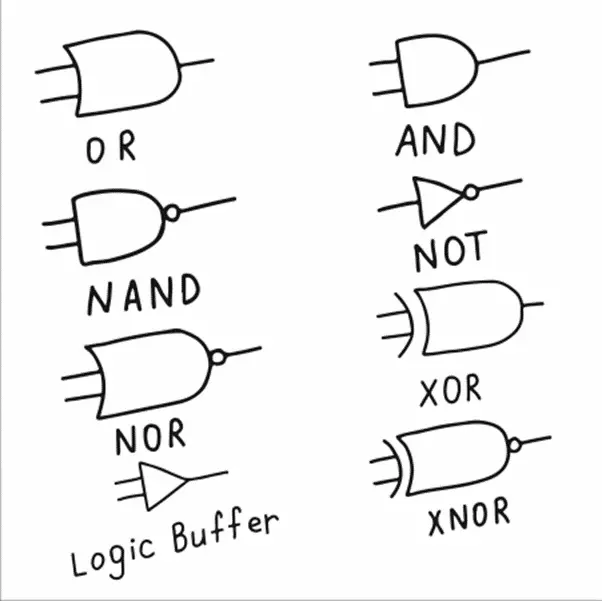








































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































0