Table of Contents
How to Give a Systematic Name to a Compound
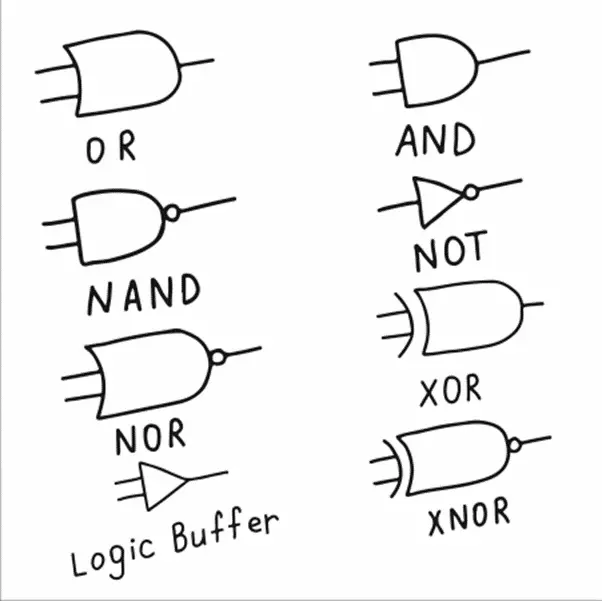
To give a name to a compound, follow the rules of IUPAC nomenclature. The compound name should be one name. The parent chain is the longest chain. Numbering should start from the side of the compound where there are more precursors. A compound’s IUPAC nomenclature should be consistent. IUPAC nomenclature follows the rules for naming ionic substances.
IUPAC nomenclature
The IUPAC nomenclature for the same compound may differ in the naming of a carboxyl group. The most common is benzoic acid. Usually, the carboxyl group is named with the suffix -oic acid. The position where the carboxyl group is located in the main chain must be designated “one”. Other compounds that have multiple functional groups must indicate which one is the main. Lower-priority functional groups are labeled as substituents. These compounds must also contain the carbon of a cyano group. The IUPAC nomenclature of the following compound follows this generalization.
The IUPAC nomenclature of the subsequent compound shows the positions of all skeletal carbon atoms in two rings. This number does not include the carbons in the side chains or the substituents over the rings. The parent chain’s carbon atoms are used to assign the locant. Since there are two different series of locants for the same compound, one must choose the right one by applying the “first-point-difference” rule.
A cyclic hydrocarbon is identified by the prefix “cyclo-“, which is derived from how many carbons are in the parent chain. The name of this compound is x,x’-bi(cycloalkyl), where x is the locant carbon in the second ring. In some cases, IUPAC nomenclature may include both formal and common names.
The IUPAC nomenclature of the subsequent compound differs in two key ways: the order of side-chains and functional groups. For example, the methyl group is listed before dihydroxy and dimethyl, while the ethylene is listed before ethyl. The tetra-methyl and terti-methyl groups are also cited.
The parent chain is typically a single atom. However, sometimes a double- or triple-bonded bond is a double- or triple-bonded bond. The name must include the IUPAC prefix to indicate what type or bonding is present. It should also indicate whether the parent chain has side chains or substituents. In addition, the parent chain must have a functional group. If there is no functional groups, the IUPAC nomenclature must indicate the presence of an element such as a substitute or a ligand.
Rules for naming compounds
When naming compounds, the first element in the compound goes first. If the compound contains only one element, the mono-prefix is not used. The same applies to compounds that have two adjacent vowels. For example, compounds with oxygen have carbon first. A compound with a single atom of oxygen is called nitric oxide. The proper name for nitrous oxide, is nitrogen monoxide. Other common names for nitrogen compounds are nitrous oxide and dinitrogen monoxide.
A compound can have multiple “branches”, i.e. A ring is a carbon atom attached to a ring. For example, pentane can have a CH3 atom attached to its second carbon. The CH3 atom is known as the methyl or ethyl group. In either case, the compound namer must include the fact of the branch and ensure that the branch is not longer than the original chain.
The names of chemical compounds follow a similar pattern as the names of geometric shapes. Tri-chloride, for example, has three chlorine atoms. The pentagon, hexagon and octagon can also be used to denote compounds that have more than one atom. A rational nomenclature system should identify chemical groups and their locations. In addition, the chemical name should indicate whether or not a compound has any functional groups, and should indicate the amount of hydrogen.
One of the easiest rules for naming molecular compounds is to write their names. Next, use the Roman numeral for the second element. The second element should be referred to as -ide. The same goes for naming polyatomic ions. If a compound is composed of more than one element, the prefix “o” is used to indicate that second non-metal.
The names of binary and ionic compounds follow the same rules as ionic compounds. The cation is the first element. The anon is the next element. The Roman numeral is used to determine the charge. Similarly, the charge of a metal ion is indicated by the number of elements present in the compound. For example, iron chloride, or FeCl2, is ambiguous because it does not distinguish between the two compounds.
IUPAC nomenclature for ionic compounds
The IUPAC nomenclature allows for the naming of inorganic chemical compounds. It was created by the International Union of Pure and Applied Chemistry and published in The Nomenclature of Inorganic Chemistry. This nomenclature lists all the elements and their compounds. These elements can all be found in nature, but they are not in the perfect world.
There are a couple of things to note about the IUPAC nomenclature of ionic compounds. First, a lot of metallic elements are known to form different positive charges. A compound that contains one of these elements must have an indication of its charge. This older method uses suffixes to indicate the higher and lower charges. For example, copper is known as Cu2+, while iron is given the name of Fe3+.
Ionic compounds have a specific structure and are named according to systematic procedures. The simplest ionic compounds are those which consist of just one type of cation or anion. Their nomenclature is trivial. To name them, the first part of the name is a cation, then an ion, and finally a suffix -ide. A diatomic compound is one that contains more than two ions.
Another thing to note is the oxidation state of the element in which the ion is present. Fe2+ in the case of iron would be FeCl2. The Roman numeral following the cation would be iron (II), chloride. If the ion is paired with oxygen, it would have the name Iron(III) chloride. However, iron(III) oxide would have the suffix -io.
Acids are another important class of compounds. An acid is named using a prefix such as hydro-. Usually, the suffix is -ic. In contrast, an oxyacid is named with an -ous or -ic. Examples of compounds that have acidic properties include hydrochloric acid and carbonic acid. This is how IUPAC nomenclature is used for ionic substances.
IUPAC nomenclature for hydrates
Hygroscopic compounds are able to absorb water from the atmosphere. They are used as solvents and dry cleaning agents. Not all hydrates can be considered hydrates. Some, like solid NaOH, absorb water from the air and then evaporate. This substance is then referred to as anhydrous. Similar to the above, many carbohydrates can release water when heated, but they are not truehydrates.
Hydrates are any chemical compound that contains water and one of its constituent elements. Hydrates contain two Na+ ions and an ion of chlorine, which is in group 17 and carries a 1-charge. In addition, hydrates can be classified based on the ion that is incorporated in the compound. When hydrated, cobalt(II), chloride turns red.
Hydrates generally contain water in a stoichiometric proportion. The name of an anhydrous chemical is used to give the ionic hydrate its name. Generally, hydrates are named with the name of their anhydrous component, followed by a prefix denoting how many moles of water they contain. Hence, washing soda is a common name for sodium carbonate hydrate, while decahydrate is the systematic name of sodium carbonate hydrate.
The IUPAC nomenclature of hydrates follows a common format. The first element in the hydrate’s name is the ionic compound, followed by a numerical suffix. The second element, or anhydride, is the water-deficient compounds. The last element is a hydrogen atom. Anhydride is a substance that has lost water. Anhydrous substances are anhydrous.
Besides ammonia, methane and chlorine are also common examples of hydrates. The most common form of methane hydrates is dihydrogen monoxide, which has a loose water framework around gas molecules. They are found in abundance in the ocean floor and on land in permafrost. Scientists believe that methane hydrates have more energy than the world’s fuel reserves.
Another type of hydrate that you might consider is sodium bicarbonate. The bi-prefix suggests the presence of an acidic hydrogen, so sodium bicarbonate is sodium hydrogen carbonate. Another form is sodium bicarbonate, which is sodium hydro sulfite. A covalent compound, on the other hand, is made of two nonmetals that share electrons. The covalent bond holds two nonmetals together.














































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































0