In a list, place these hydrocarbons in order of decreasing or increasing boiling point. They should be listed from highest to lowest, from hottest to coldest. Items should overlap to be considered equivalent. If they do not, then they are not equivalent. If they do, they are not comparable. However, you can use this information to compare the properties of different substances. Here are some tips for placing different hydrocarbons in order of decreasing boiling point:
The length of a carbon chain is directly related to the boiling point of a compound. The longer the chain, the higher the boiling point. The longest hydrocarbons are paraffin, which has the most carbon atoms, and ethane has just two. The surface area of a compound also influences its boiling point. The larger the surface area, the more intermolecular forces will occur.
The length of a carbon chain is directly related to the boiling point of a hydrocarbon. The longer the carbon chain, the higher its boiling point. The shortest hydrocarbon, methane, has two carbon atoms. The surface area of a compound is also related to the boiling point. A bigger surface area means stronger intermolecular forces, and a higher boiling point.
The boiling points of hydrocarbons depend on the number of carbon atoms. The greater the number of carbon atoms, the higher the boiling point. Similarly, the larger the surface area, the higher the boiling point. As a result, these compounds have high-quality surfaces. The greater the surface area, the lower the boiling point. If the surfaces of different substances are too large, it can cause cracking.
The boiling point of a hydrocarbon is related to the length of the carbon chain. The more carbon atoms there are, the higher the boiling point. The length of the chain determines the boiling point. This is important, as it will determine the chemical properties of the substance. Moreover, the length of the carbon chain affects the boiling point. When a gas is heated over a flame, the boiling points of the gas will increase.
The boiling point of a hydrocarbon is the temperature at which the vapor pressure equals atmospheric pressure. The vapor pressure of a hydrocarbon depends on the amount of carbons and branching in the chain. Its average kinetic energy is enough to overcome the forces of attraction between two molecules. As a result, a liquid boils when it reaches its boiling point. This means that the volume of a gas is equal to the pressure of the surrounding air.
If you want to know which hydrocarbons have the highest boiling point, then you need to consider the type of heating element they possess. The highest boiling point of oil is at approximately 445°F. The lowest is at 290°F. When hot, it has a lower boiling point than hydrogen. In fact, it’s just about as hot as water. And it’s a bit more difficult to heat a gasoline-powered vehicle at these temperatures.
The boiling point of a hydrocarbon varies according to the number of carbon atoms in its chain. The more carbon atoms a hydrocarbon contains, the higher its boiling point will be. The highest boiling point is paraffin, while ethane contains only two carbon atoms. The lowest boiling point is ethane, which has the least number of carbon atoms.
Hydrocarbons are classified according to their boiling point. Their melting points vary depending on the chain length. The highest melting point is paraffin, while the lowest is ethane, which is composed of two carbon atoms. The boiling point is also affected by the surface area of a hydrocarbon. The larger the surface area of a compound, the higher its boiling temperature. This means that a substance will have a lower melting point than hydrogen.











































































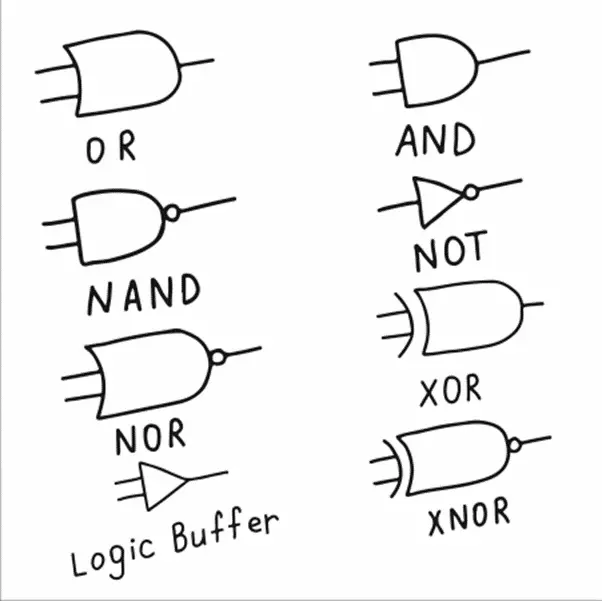





































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































0