Table of Contents
Hitech Industries’ Research and Development Wing
Hitech Industries has a well-defined research and development roadmap. Its R&D wing comprises of scientists from various disciplines who are continuously engaged in innovative ideas and researches. The R&D wing’s aim is to develop new synthetic resins and chemicals and find innovative uses for them. This helps the company meet the specialized requirements of its global customers. It is manned by highly-qualified staff and is backed by state-of-the-art equipment.
HITECH Act
The Health Information Technology for Economic and Clinical Health (HITECH Act) set ambitious goals to improve health care delivery through the use of electronic health information. While the law has been largely effective in achieving these goals, many questions still remain. This article looks at the first five years of HITECH Act implementation, assesses the challenges still remaining, and considers lessons learned.
The legislation outlines guidelines to ensure the security and privacy of health information and the implementation of health information technology. It also requires that healthcare providers conduct security audits to ensure that they meet certain minimum standards. It also requires them to implement a security program that meets HIPAA privacy requirements. As a result, HITECH Act research is expected to focus on the impact of this legislation on health care organizations.
Subtitle B–Testing of Health Information Technology
Testing of health information technology is an essential component of health information technology. This act imposes requirements for testing health information technology, including the use of standards and criteria for the use of health information technology. In addition, it establishes the National Coordinator for Health Information Technology, who maintains a website that is regularly updated and provides information on current activities, reports, and recommendations.
A regional center for health information technology is a nonprofit organization affiliated with an institution in the United States or a group of nonprofit organizations. It must meet criteria for certification and receive funding based on merit. A regional center shall provide technical assistance and disseminate information about health information technology to improve healthcare. The activities of the regional center shall be in accordance with the National Coordinator’s strategic plan.
Subtitle C–Funding for Health Information Technology
The HITECH Act was signed into law on February 17, 2009. The new law aims to improve healthcare by promoting the adoption of health information technology (health IT). The act includes two parts, Part 1 focuses on advancing the use and application of health information technology standards and Part 2 concerns the privacy and security of electronic health information. The HITECH Act also strengthens the enforcement of HIPAA, which has been criticized for failing to protect patient health information. The legislation also includes companion breach notification regulations for non-HIPAA-covered organizations.
The Act defines the cost of medical care as “any costs associated with the direct provision of health care” and “the making of grants for the direct provision of health care services.” The Act also defines a “protected health information” (PHI) as information about a single individual.
Penalties for violations of HIPAA
Penalties for violations of HIPAA, the federal privacy law, can range from a few thousand dollars to a million dollars. The penalties depend on the nature of the violation, the number of affected individuals, and the financial situation of the covered entity. The goal of HIPAA penalties is to punish covered entities that violate the law without crippling their operations. In addition, HIPAA penalties are meant to send a strong message to other healthcare organizations.
In recent years, several states have stepped in to enforce HIPAA penalties. Massachusetts, Indiana, Vermont, Minnesota, and Connecticut have all acted to hold companies responsible for HIPAA violations. These new laws may lead to more fines in the future. Additionally, individuals responsible for violations may lose their jobs and face criminal charges.
Willful neglect penalty under HITECH
The HITECH Act, which was included in the American Recovery and Reinvestment Act of 2009, includes a new penalty for “willful neglect” violations. The penalty is a monetary penalty that is imposed upon healthcare entities that fail to comply with the new laws. These penalties start at $50,000 for each violation.
The HITECH Act is a new law that aims to improve the security and privacy of health information. The new legislation strengthens government enforcement and establishes penalties for violations of HIPAA. A willful neglect violation can bring a civil money penalty of up to $1.5 million.
The HITECH Act contains new penalties for willful neglect of HIPAA. The maximum penalty for willful neglect is now $250,000 per violation, and for a repeat violation, $1.5 million. The new penalty structure also expands HIPAA’s penalty scheme to business associates. Business associates must get assurances that they will protect patient information.







































































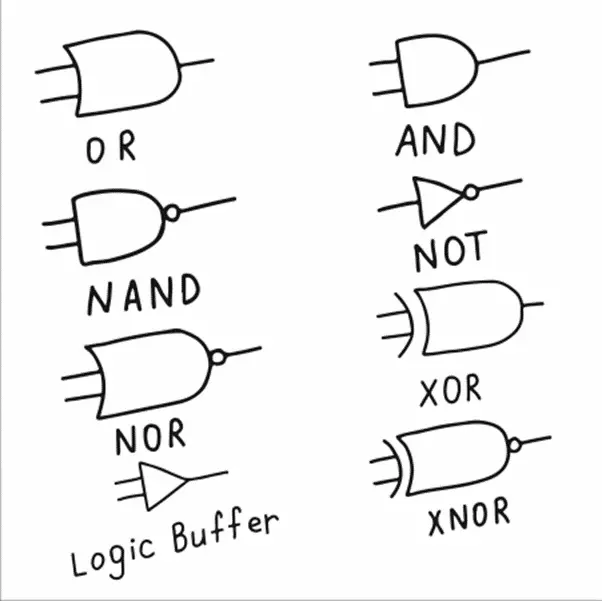







































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































0