The question in step two asks us to draw the major organic product of the reaction shown. We need to draw a diagram containing all the charges that take place in the reaction. Then we need to write down the product(s) of the reaction. In step three we have to write down the main products of the reaction. Hence we need to draw the product of the reaction. In step four we have to draw the product of the reaction.
Let’s start by drawing the reaction. The main organic product of the reaction shown is 2-methyl-2-pentanol. The minor product is 1-propanol. The amines form both the major and minor organic products. In the case of the major organic product, the heat produced during the process leads to the formation of a carbonyl compound. The reaction ends in a KOC(CH3)3 CH3 H2SO4.
In each reaction, draw the corresponding ions. In the case of 2-methyl-2-pentanol, the amine structure should be modified to produce the corresponding ammonium chloride salt. In each step, place asterisks next to the chirality centers of the amine. The final step is to draw the minor organic product of the reaction shown. The reaction is called oxidative dehydrogenase.
When the amine and the electrophile react, an ortho- and a para-position is obtained. In the second step, the bromine atom reacts with the amine to form a cyclic compound of 2-methyl-2-pentanol. The resulting oxidation produces an alcohol-carbon-based amide. A common reaction is a hydrogen-bonding reaction.
In step two, the amine is oxidized by the Dess-Martin reagent and the major organic product is KOC(CH3)3 CH3 H2SO4 (Mep-CH3CH3P). The major organic product of the reaction shown is MCP(CH3)3 CH3CH3CH3H2SO4. Then, the bromine atom reacts with the amine, and produces the products.
Visit the rest of the site for more useful articles!

































































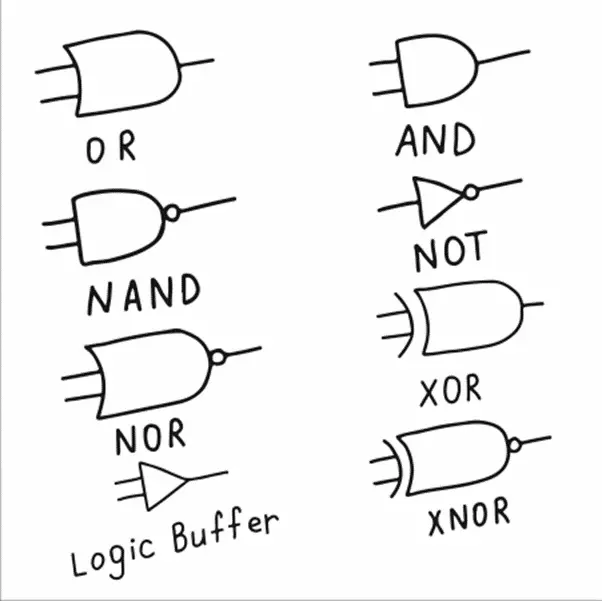



































































































































































































































































































































































































































































































































































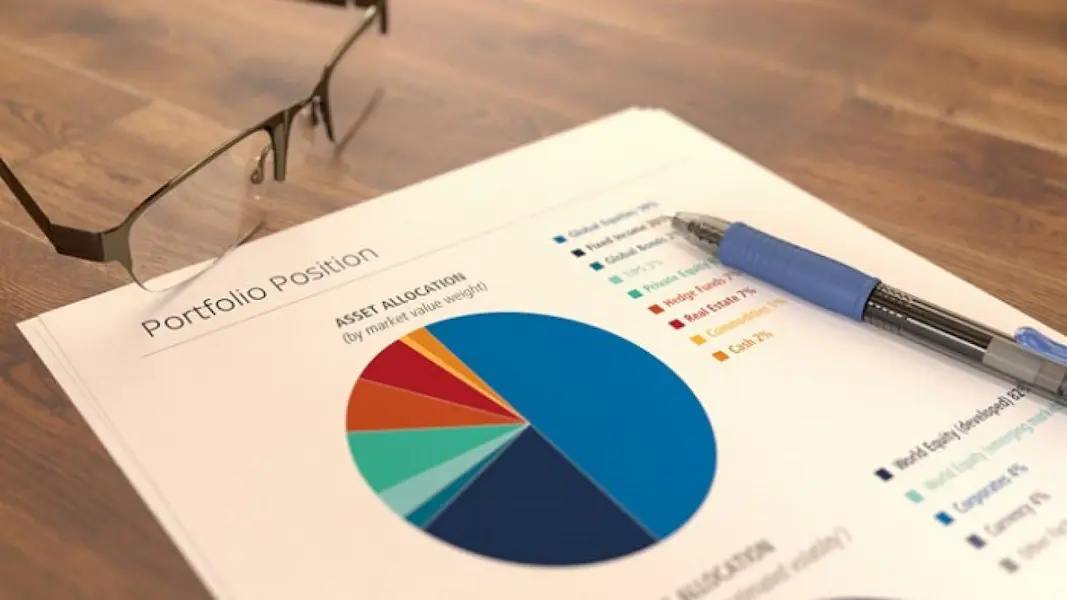








































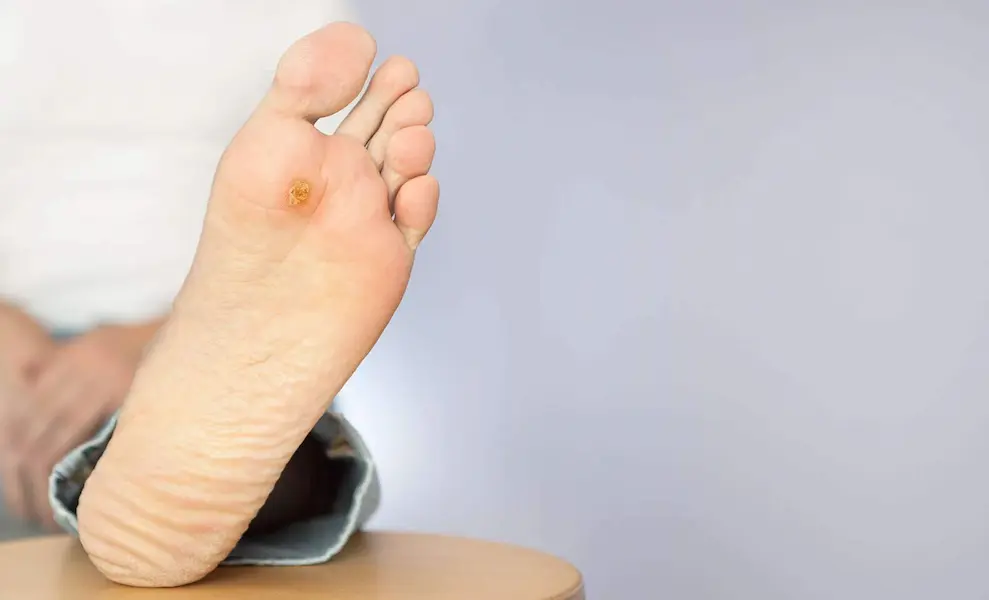

































































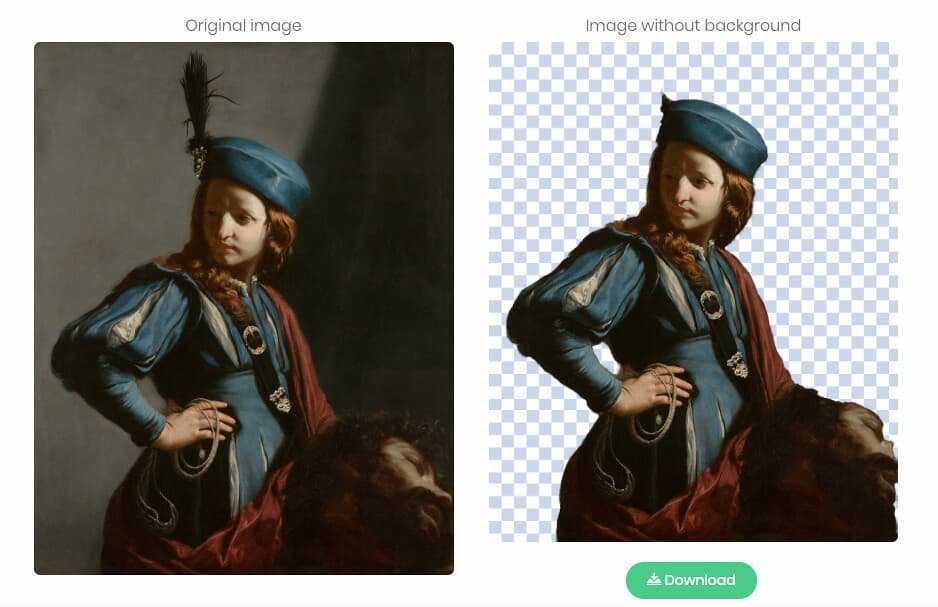






























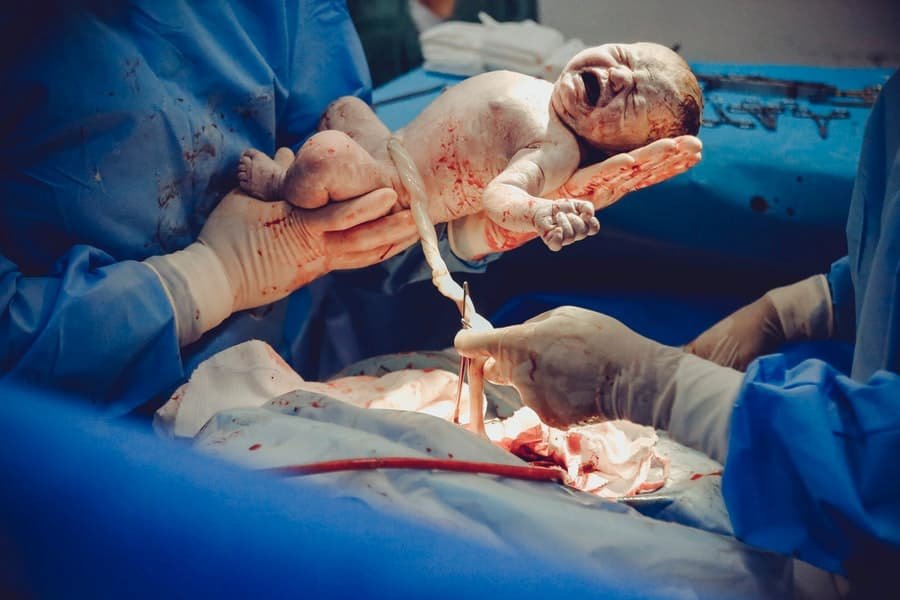






























































































































































































































































































0